7. THE p-BLOCK ELEMENTS – LONG ANSWER TYPE QUESTIONS
7. THE p-BLOCK ELEMENTS
Q. 1. Why does the reactivity of nitrogen differ from phosphorus ?
Ans⇒ Nitrogen molecule is a diatomic and the two nitrogen atoms are linked by triple bond (N = N). As the bond dissociation energy is very high (946 kJ mol-1). It is not possible to cleave the triple bond so easily. Therefore the reactivity of nitrogen differ from phosphones.
Q. 2. Why does NH3 form hydrogen bond but PH3 does not ?
Ans⇒ N-H bond is reasonable polar and this leads to hydrogen bonding As the bond polarity of the P-H bond is almost negligible. PH3 is not involved in hydrogen bonding.
Q.3. How is ammonia manufactured industrially ?
Ans⇒ Ammonia is manufactured industrially by Haber’s process.
N2 (g) + 3H2 (g) ⇌ 2NH3 (g)
Δf = -46.1 kJ mol-1 HO
A mixture of dry nitrogen and hydrogen gases in the ratio of 1 : 3 by volume is compressed to about 200 to 300 atm and passed over iron mixed with aluminium oxide (Al2O3) and potassium oxide (K2O) which act as promotors.
Ammonia being formed is continuously removed by liquefy it.
The optimum conditions for the production of ammonia are a pressure of about 200 atm., a temperature of – 700 K and use of a catalyst such as iron oxide with small amounts of K2O and Al2O3 to increase the rate of attainment of equilibrium.
Q. 4. How are XeO3 are XeOF4 prepared ?
Ans⇒ (i) Preparation of XeO3 : It is prepared by the hydrolysis of XeF4 and XeF6 under controlled pH of medium.
6XeF4 + 12H2O → 4Xe + 2XeO3 + 24HF + 3O2
XeF6 + 3H2O → XeO3 + 6HF
(ii) Preparation of XeOF4 :
Partial hydrolysis of XeF6 gives XeOF4
XeF6 + H2O → XeOF4 + 2HF
Q. 5. Why do noble gases have comparatively large atomic sizes ?
Ans⇒ The atomic radii of noble gases are called Vander Waals radii and therefore these are exceptionally higher than theri correspoding adjacent members of same period because for them either covalent radii or ionic radii are determined.
Q. 6. List the uses of neon and argon gases.
Ans⇒ Uses of Neon : (i) Neon is mainly used in fluorescent lamps of tubes for advertising purposes. These are known as neon signs and can be seen at long distances even when there is a fog. Neon actually produces an orange red glow in the tube and on mixing with the vapours of other gases, glows or signs of different colours can be obtained.
(ii) It is used in filling sodium vapour lamps.
(iii) It is used in safely devices for protecting certain electrical instruments (voltmeters, relays, rectifiers.)
Uses of Argon : (i) It is used in metal filament electric lamps since it increase the life of the tungsten filament by retarding its vapourisation.
(ii) A mixture of argon and mercury vapoursis used in fluorescent tubes.
(iii) It is used to create an inert atmosphere for welding and for carrying certain chemical reactions.
Q. 7. Assign reason for each of the following :
(a) Noble gases are mostly chemically inert.
(b) Bismuth is a strong oxidizing agent in a pentavalent state.
Ans⇒ (a) Noble gases are mostly chemically inert due to the following reasons :
(i) Atoms of the noble gases have stable closed shell electronic configuration.
(ii) Noble gases have exceptionally high ionisation energies.
(iii) Noble gases have very low electron affinities.
(b) The stability of + 5 oxidation state decreases down the group due two inert pair effect. So +5 oxidation state of Bi is ess stable than its + 3 oxidation state.
Q. 8. NF3 does not have donor properties like ammonia. Explain.
Ans⇒ NF3 has a pyramidal shape with one lone-pair on N atom.
The lone-pair on N is in opposite direction to the N-F bond moments and therefore it has very low dipole moment Gohout 0.234 D). Thus it does not show donor properties. But ammonia has high dipole moment because lone pair is in the same direction as the N-H. bond moments. Thus it has donor properties
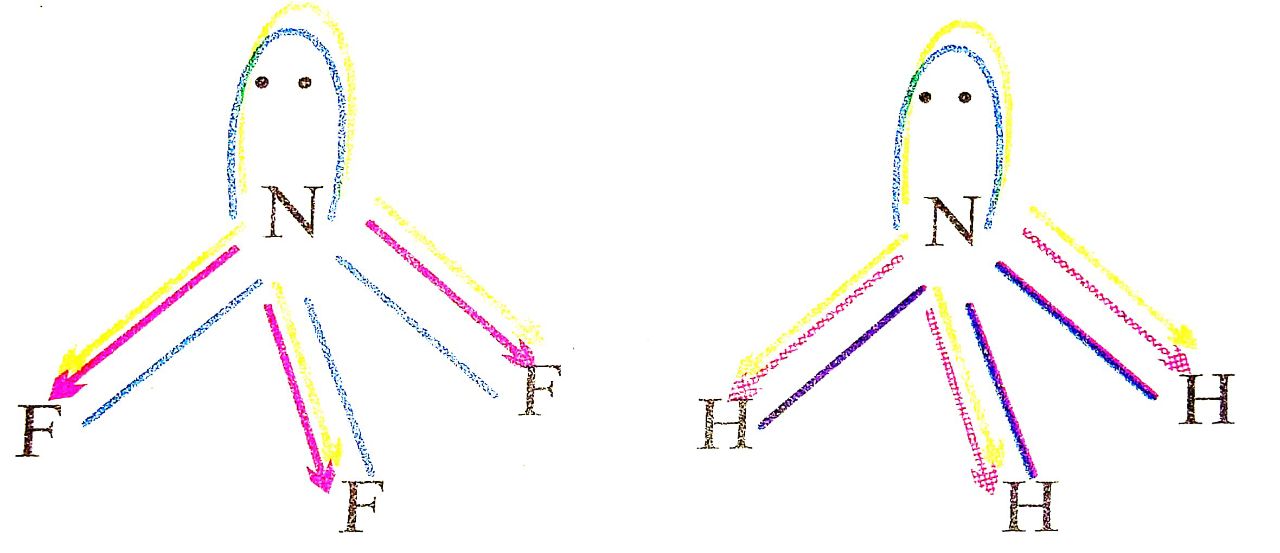
Q. 9. Describe the chief uses of fluorine, chlorine and their compounds.
Ans⇒ Uses of fluorine :
(i) It is used in the preparation of fluoro carbon which are non-inflamable and chemically inert and are used as solvents, lubricants and insulator.
(ii) It is used as a refrigerant in many of the cooling processes.
(iii) It finds considerable use as DDFT, which similar to DDT, is extremelly efficient as a fun gicide and fumigant.
(iv) In nuclear physics and higher voltage electricity, fluorine as SF, finds great use e.g., in the separation of isotopes of uranium.
Uses of chlorine :
(i) It is used in sterlization of drinking water.
(ii) Large quantities of chlorine are used for bleaching paper pulp and textiles.
(iii) It is used in the manufacture of inorganic chemicals such HCl. Sodium hypochlorite (NaOCl), bleaching power (CaOCl2), phosphorus trichloride (PCl3), phosphorus pentachloride (PCl5) etc.
(iv) It is used in the manufacture of vinyl chloride which is the starting material for the plastic polyvinyl chloride (PVC).
(v) It is used in the manufacture of insecticides like DDT. germicides, dyes and drugs.
Q. 10. Discuss the general characteristics of Group 15 elements with reference to their electronic configuration, oxidation state, atomic size, ionisation enthalpy and eletronegativity.
Ans⇒ Electronic configuration :
All the elements of Group 15 have ns2np3 (= 5 electrons) electronic configuration in their valence shells. The s-subshell is completely filled and p-subshell is
exactly half-filled (P’xP’yP’z). This imparts extra stability to their electronic configuration.
Nitrogen = N = 7 = [He] 2s2p3
Phosphorus = P = 15 = [Ne] 3s23p3
Arsenic = As = 33 = [Ar] 3d104s24p3
Antimony = Sb = 51 = [Kr] 4d105s25p3
Bismuth = Bi = 83 = [Xe] 4ƒ 145d106s26p3
Oxidation States : The common oxidation states of these are – 3, + 3 and + 5. The tendency to show -3 oxidation state decreases from top to bottom ase in size and metallic character. N does idation state because it has only 4 orbitals
(One s and three p) for bonding. Br hardly forms any compound in the – 3 oxidation state. The stability of + 5 oxidation state decreases down the group and that of + 3 state increases (due to inert pair effect). N exhibits + 1, +2 and + 4 oxidation states when it reacts with oxygen. P also shows + 1 and + 4 oxidation states in some oxoacids.
Atomic size : Atomic size increases from N to Bi due to the addition of one orbit each time. There is a considerable increase in covalent radius from N to P. However from As to Bi, only a small increase in covalent radius is observed due to the presence of completely filled d and / or ƒ orbitals in heavier members.
Ionisation Enthalpy : The ionisation enthalpy of group 15 elements is much greater than that of Group 14 elements due to the extra-stability of half-filled p orbitals and smaller size. It decreases from top to bottom due to gradual increase in atomic size.
Electronegativity : Electronegativity decreases from top to bottom in the group. The difference of electronegativity is not much pronounced amongst the heavier elements.
Class 12th Chemistry Long Type Question
| S.N | CHEMISTRY LONG TYPE QUESTION 2022 |
| 1 | SOLID STATE |
| 2 | SOLUTION |
| 3 | ELECTROCHEMISTRY |
| 4 | CHEMICAL KINETICS |
| 5 | SURFACE CHEMISTRY |
| 6 | GENERAL PRINCIPLES AND PROCESSES OF ISOLATION OF ELEMENTS |
| 7 | THE P-BLOCK ELEMENTS |
| 8 | THE D- AND f-BLOCK ELEMENTS |
| 9 | CO-ORDINATION COMPOUNDS |
| 10 | HALOALKANES AND HALOARENES |
| 11 | ALCOHOLS, PHENOLS AND ETHERS |
| 12 | ALDEHYDES, KETONES AND CARBOXYLIC ACIDS |
| 13 | AMINES |
| 14 | BIOMOLECULES |
| 15 | POLYMERS |
| 16 | CHEMISTRY IN EVERYDAY LIFE |
| 17 | DISTINGUISH BETWEEN PAIR |
| 18 | CONVERSIONS |



